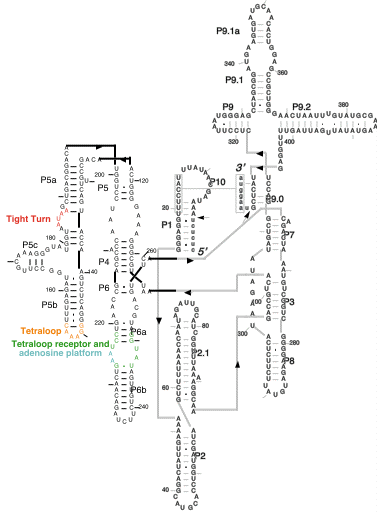
The group I intron from Tetrahymena was the first RNA for which a catalytic activity was described (ribozyme). It consists of ~400 nts of RNA, shown below, which can self-splice itself from a ribosomal RNA transcript.
Reference:
Cate, et al., 1996. Crystal Structure of a Group I Ribozyme Domain: Principles of RNA Packing. Science 273:1678. [Medline abstract] Full-text HTML version
I. Introduction
The group I intron from Tetrahymena was the first RNA for which
a catalytic activity was described (ribozyme). It consists of
~400 nts of RNA, shown below, which can self-splice itself from
a ribosomal RNA transcript.
The crystal structure of the entire ribozyme has not been determined.
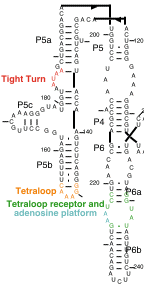
However, a domain of the intron, which has been shown to fold
autonomously, called P4-P5-P6, has been crystallized.
Click here <
I. The tetraloop receptor
The GNRA tetraloop is a conserved 4 nucleotide loop which is commonly
found on RNA helices. In a previous theoretical model of the groupI
intron structure, Michel
and Westoff noticed that the sequence of some GNRA tetraloops
influenced the sequence of base pairs in other helices, suggesting
that they might form a tertiary interaction. Costa
and Michel further noticed that the GAAA tetraloop seemed
to interact with a particular sequence motif, termed the tetraloop
receptor. Click here to see the structure of the tetraloop and
receptor <
II. The adenosine platform
Residues A225 and A226 within the tetraloop receptor form a structure
termed the adenosine platform. These consecutive residues H-bond
with each other (from N3 of A225 to N6 of A226) to form a motif
which presents the adenosine residues for H-bonding or stacking
with other residues. To zoom in on the adenosine platform of the
tetraloop receptor, click here <
III. The tight turn
Residues 183 through 188 of the P4-6 domain form a tight turn
of the RNA backbone. The phosphate oxygens of the backbone residues
come within 3 angstroms in places. This is made possible by two
Mg++ cations which coordinate with the phosphate oxygens of residues
183, 184, 186 and 187. Click here to zoom in on the tight turn
<
![]() Back
to intro to DNA-RNA structure.
Back
to intro to DNA-RNA structure.
![]() This section Still Under Construction
This section Still Under Construction