Earth is covered by a thin “veneer” of sediment.
The veneer caps igneous and metamorphic “basement.”
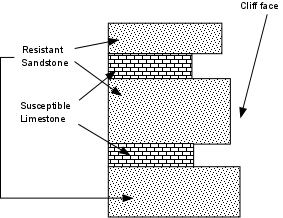
This sediment cover varies in thickness from 0 to 20 km. It is thinner (or missing) where igneous and metamorphic rocks outcrop, and is thicker in sedimentary basins.
In order to make this sediment and sedimentary rock, several steps are required:
- Weathering – Breaks pre-existing rock into small fragments or new minerals
- Transportation of the sediments to a sedimentary basin.
- Deposition of the sediment
- Burial and Lithification to make sedimentary rock.
Each Step in the process of forming sediment and sedimentary rocks leaves clues in the sediment. These clues can be interpreted to determine the history of the sediment and thus the history of the Earth.
Weathering
Geologists recognize two categories of weathering processes
- Physical Weathering
- disintegration of rocks and minerals by a physical
or mechanical process.
- Chemical Weathering
- chemical alteration or decomposition of rocks and
minerals.
Although we separate these processes, as we will see, both work together to break down
rocks and minerals to smaller fragments or to minerals more stable near the Earth's
surface.Both types are a response to the low pressure, low temperature, and water and oxygen rich nature of the earth’s surface.
Physical Weathering
The mechanical breakup or disintegration of rock doesn't change mineral makeup. It creates broken fragments or “detritus.” which are classified by size:
- Coarse-grained – Boulders, Cobbles, and Pebbles.
- Medium-grained – Sand
- Fine-grained – Silt and clay (mud).
Physical weathering takes place by a variety of processes. Among them are:
- Development of Joints - Joints are regularly spaced fractures or
cracks in rocks that show no offset across the fracture (fractures that show an
offset are called faults).
- Joints form as a result of expansion due to cooling or relief of pressure as overlying
rocks are removed by erosion.
- Igneous plutons crack in onion like “exfoliation” layers.
These layers break off as sheets that slide off of a pluton.
Over time, this process creates domed remnants. (See figure B.4 in your text)
Examples: Half-Dome (CA.) (see figure 22.12a in your text) and Stone Mountain (GA.).
- Joints form free space in rock by which other agents of chemical or physical weathering
can enter.
- Crystal Growth - As water percolates through fractures and pore spaces it may contain
ions that precipitate to form crystals. As these crystals grow they may exert an
outward force that can expand or weaken rocks.
- Thermal Expansion - Although daily heating and cooling of rocks do not seem to have an effect,
sudden exposure to high temperature, such as in a forest or grass fire may cause expansion
and eventual breakage of rock. Campfire example.
- Root Wedging - Plant roots can extend into fractures and grow, causing expansion of the fracture.
Growth of plants can break rock - look at the sidewalks of New Orleans for
example.
- Animal Activity - Animals burrowing or moving through cracks can break rock.
- Frost Wedging
- Upon freezing, there is an increase in the volume of the
water (that's why we use antifreeze in auto engines or why the pipes break in New Orleans
during the rare freeze). As the water freezes it expands and exerts a force on its
surroundings. Frost wedging is more prevalent at high altitudes where there may be
many freeze-thaw cycles.
|