| EENS 2120 |
Petrology |
| Prof. Stephen A. Nelson |
Tulane University |
|
Igneous Rocks of the Convergent Margins |
|
| The convergent plate margins are the most intense areas of active magmatism above sea
level at the present time. Most of world's violent volcanic activity occurs along
these zones. In addition, much magmatism also has resulted (and probably is
resulting at present) in significant additions to the crust in the form of plutonic
igneous rocks. Here, we look at this magmatism in terms of the volcanic rocks
that appear to be related to subduction. Occurrence |
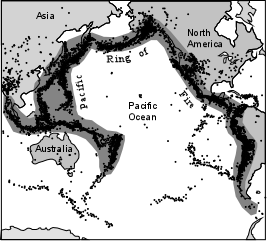
| The "Pacific Ring of Fire" is often discussed in relation to both earthquakes and volcanoes. It surrounds the Pacific Ocean basin and extends into the Indian Ocean and Caribbean Sea. Active subduction is taking place, along these convergent plate boundaries, as evidenced by the zone of earthquakes, called a Benioff Zone, that begins near the oceanic trenches and extends to deeper levels in the direction of plate motion. Earthquake focal depths reach a maximum of about 700 km in some areas. Volcanism occurs on the upper plate about 100 to 200 km above the Benioff Zone. For this reason, volcanism in these areas is often referred to as subduction-related volcanism. |  |
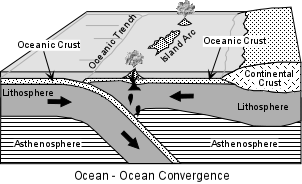
Two situations occur.
|
 |
|
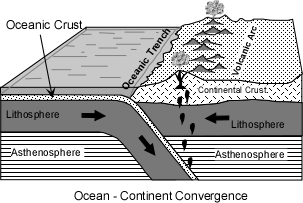 |
| Within these volcanic arcs the most imposing, and therefore most recognized by early workers, features of the landscape are large stratovolcanoes. These usually consist of predominantly andesitic lava flows and interbedded pyroclastic material. But, in the late stages of volcanism more silicic lavas and pyroclastics like dacites and rhyolites are common. |
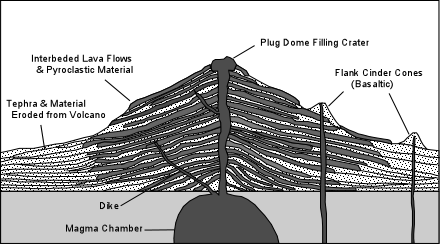 |
| Many of these stratovolcanoes pass through a stage where their upper
portions collapse downward to form a caldera. These caldera forming events are usually associated with
explosive eruptions that emit silicic pyroclastic material in large-volume eruptions.
It is the sudden evacuation of underlying magma chambers that appears to result in
the collapse of the volcanoes to form the calderas. The imposing presence of these large mostly andesitic stratovolcanoes led to an early widespread perception among petrologists that basalts were rare or absent in these environments. In recent years, however, it has become more evident that basalts are widespread, but do not commonly erupt from the stratovolcanoes. Instead, they are found in areas surrounding the stratovolcanoes where they erupt to form cinder cones and associated lava flows. One explanation for this distribution is that the magma chambers underlying the stratovolcanoes intercept the basaltic magmas before they reach the surface and allow the basalts to differentiate to more siliceous compositions before they are erupted. Basaltic magmas that are not intercepted by the magma chambers can make it to the surface to erupt in the surrounding areas. |
Petrography Probably the most distinguishing feature of subduction-related volcanic rocks is their usually porphyritic nature, usually showing glomeroporphyritic clusters of phenocrysts. Basalts commonly contain phenocrysts of olivine, augite, and plagioclase. Andesites and dacites commonly have phenocrysts of plagioclase, augite, and hypersthene, and some contain hornblende. The most characteristic feature of the andesites and dacites is the predominance of fairly calcic plagioclase phenocrysts that show complex oscillatory zoning. Such zoning has been ascribed to various factors, including:
|
Rhyolites occur as both obsidians and as porphyritic lavas and pyroclastics. Phenocrysts present in rhyolites include plagioclase, sanidine, quartz, orthopyroxene, hornblende, and biotite. In addition to these features, petrographic evidence for magma mixing is sometimes present in the rocks, including disequilibrium mineral assemblages, reversed zoning etc. Xenoliths of crustal rocks are also sometimes found, particularly in continental margin arcs, suggesting that assimilation or partial assimilation of the crust could be an important process in this environment. Major Elements |
Before discussing the major element chemistry of subduction related volcanic rocks we first need to clarify some terminology concerning rock suites. In the early 1900s a petrologist by the name of Peacock examined suites of rocks throughout the world. On a plot of CaO and total alkalies versus SiO2, Peacock noted that the two curves intersected at different values of SiO2 for different suites. He used the value of SiO2 where the two curves intersect (now known as the Peacock Index or Alkali-Lime Index) to divide rock suites into the following: |
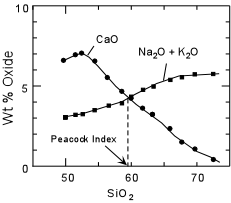 |
Peacock Index Name of Suite <51 Alkalic 51-56 Alkali-Calcic 56-61 Calc-Alkalic >61 Calcic
| Although Peacock's classification of rock suites is rarely used today, some of the terminology has survived in slightly different forms. For example the general term "alkaline suite" is used to describe rock suites in which the basic rocks have relatively high values of total alkalies, like the alkali basalt -hawaiite - mugearite - trachyte suite or the basanite - nephelinite suites discussed previously. Most subduction related volcanic and plutonic rocks fall into the calc-alkalic suite of Peacock, and thus the term calc-alkaline is often given to the suite of rocks found associated with subduction. But, it is notable that all four suites of rocks defined by Peacock are found in subduction-related areas. |
| The modern definition of the calc-alkaline suite is based on the AFM diagram. On this diagram subduction-related volcanic rocks show a trend along which the ratio of MgO to total iron (MgO/FeO*) remains nearly constant. This trend is often referred to as the Calc-Alkaline trend. Note that the calc-alkaline trend is distinct from the Fe-enrichment trends shown by the alkaline and tholeiitic suites discussed previously. Also, calc-alkaline basalts, like tholeiitic basalts are subalkaline, but they differ from tholeiitic basalts in their higher concentrations of Al2O3, with values of 17 to 20 wt %. Thus, these calc-alkaline basalts are often referred to as high alumina basalts. | 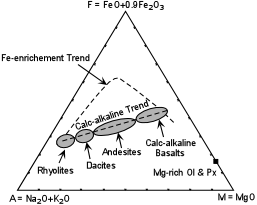 |
Recall that the Fe-enrichment trend exhibited by the tholeiitic and alkaline rock
series can be explained by crystal fractionation involving removal of early crystallizing
Mg-rich olivines and pyroxenes from the parental basaltic magmas. The calc-alkaline
trend, however, would require early removal of mineral assemblages with a higher Fe/Mg
ratio, or some other process. Over the years several explanations for the
calc-alkaline trend have been discussed. Among these are:
In recent years more light has been shed on the possible origin of the calc-alkaline suite. Perhaps the best evidence comes from experimental petrology and recent advances in experimental techniques. Experimental petrology has long suffered from the possibility that the experimental charge could possibly react with the container in which it was placed. Thus, the choice of the container or capsule, as it is called, is very important. Perhaps the best in this regard is gold. Gold remains relative inert at high temperatures, and thus does not appear to react with silicate liquids in any major way. But, the melting temperature of Gold is about 1060oC which means that experiments must be conducted at temperatures that are relatively low compared with those of basaltic liquids. Platinum (Pt) has a much higher temperature and is inert and does not react with liquids that have no Fe. But Pt absorbs Fe from liquids if it is present. Thus, if one is attempting to determine whether or not an Fe-rich phase crystallizes from a liquid at high temperature, Fe-loss to the Pt capsule could become important (i.e. it might suppress the crystallization of an Fe-rich phase because there is less Fe in the liquid than would be present under natural conditions). Although this limitation was recognized and attempts were made in early experiments to minimize Fe loss to the Pt, the experiments still remained suspect. In the 1980's, however, techniques were developed to saturate the Pt capsules with Fe prior to experimentation. This led to important new experiments addressing the problem of the calc-alkaline suite. |
| First, however, consider experiments conducted at low pressure on tholeiitic basalt magmas. These experiments show that at low pressure Plagioclase and Olivine crystallize first, with proportion of plagioclase crystallizing being higher than that of olivine. On the projected phase diagram, removal of Olivine and Plagioclase drives the liquid composition away from the Olivine corner until it intersects the Ol + Plag + Cpx + Liq. cotectic. Further crystallization of these phases will then drive the liquid composition along the cotectic to eventually crystallize pigeonite (low Ca-pyroxene) as shown by the light colored path on the diagram. | 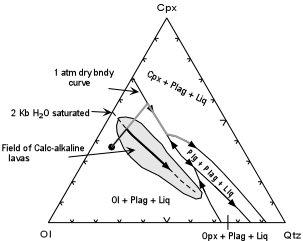 |
| Analyses of the liquids produced in these experiments showed that, as expected the
liquids would follow a trend of Fe-enrichment and thus the calc-alkaline trend could not
be produced by fractional crystallization at low pressures. Next, experiments were conducted at a pressure of 2 kb with enough H2O in the capsules to assure that the liquid would be H2O saturated at this pressure (i.e. a free vapor phase would coexist with the liquid). These experiments were conducted because it was known that H2O would lower the temperature of appearance of the silicate minerals, but would lower the temperature of appearance of oxide minerals, like magnetite to a lesser extent, and could stabilize a hydrous phases like hornblende at a higher temperature. |
The experiments show that
Furthermore, if subduction related arc rocks are plotted on the projection there are seen to lie in a field surrounding the 2 kb H2O saturated cotectic. This indicates that the calc-alkaline suite could be produced by fractional crystallization under moderate pressure water saturated conditions. This would suggest that the main difference between tholeiitic rocks and calc-alkaline rocks might be the presence (in calc-alkaline basalts) or absence (in tholeiitic basalts) of H2O in the parental magmas and/or the source rocks that melt. From our previous discussion, we know that it is possible to introduce water into the subduction related environment by dehydration of the subducting lithosphere, whereas it is more difficult to envision a mechanism to add water to the source where tholeiitic magmas are generated. |
Trace Elements As mentioned before, the trace element concentrations found in subduction related volcanic rocks are not consistent with derivation of the magmas from partial melting of subducted oceanic crust. First we will look at what the trace element concentrations show, then discuss why they are not consistent with an origin involving direct melting of the oceanic lithosphere, and then discuss how the trace element patterns of subduction related magmas might develop. As we will see the subducted oceanic lithosphere likely does make a contribution to calc-alkaline magmas, but not necessarily as the primary source of these magmas. |
| When plotted on an incompatible trace element diagram (often referred to
as a spider diagram), calc-alkaline rocks show an irregular pattern with many peaks and
valleys, unlike the relatively smooth patterns exhibited by OIBs and MORBs. Two
things of note are shown on this diagram. First, in calc-alkaline basalts (as well
as andesites, not shown) the heavy REE, represented by Yb are not depleted relative to
MORBs. |
 |
Second, the elements Nb, Ta, and Ti show negative anomalies (depletion) relative to
elements like Ba, K, La, and Ce. We'll discuss the implications of each of these
points below:
|
 |
| The addition of fluids has an additional effect, discussed earlier, in lowering the solidus temperature of the mantle and inducing partial melting. Furthermore, addition of fluids at the source of calc-alkaline magmas would explain why these magmas would become saturated with water at lower pressures to fractionate to produce the calc-alkaline trend on an AFM diagram. |
Isotopes |
|
Sr and Nd isotopic ratios for subduction-related volcanic rocks are
similar to OIBs, but show higher ratios of 87Sr/86Sr, and
extend to lower ratios of 143Nd/144Nd. Three points are notable
about this data.
|
 |
The extent of subducted sediment involvement can, in some cases be evaluated by looking at an isotope of Beryllium, 10Be, and a highly incompatible trace element Boron (B). 10Be is an isotope of Be that is produced in the upper atmosphere by bombardment of cosmic rays. Once produced it has a half-life of 1.5 million years. Be and its radioactive isotope are absorbed by the oceans and are adsorbed onto the surface of clay minerals. Because of its short half life, only small quantities 10Be remain after the passage of more than about 10 million years. If oceanic sediment is subducted and contributes material to the source of calc-alkaline magmas before it has completely decayed, then we should see small amounts of 10Be in the magmas and rocks. But note that this will only be true for very young sediment and in young volcanic rocks. So, we do not necessarily expect to see 10Be in all subduction related rocks. B is an element that is abundant in sediments but has very low concentrations in the mantle. B/Be ratios for mantle rocks are also low. Furthermore, B is much more readily soluble in fluids, so fluids derived from dehydration of the sediments have much higher B/Be ratios than the mantle and sediments. Thus, if we look at the concentration of 10Be and the B/Be ratio in subduction-related volcanic rocks we may be able to determine whether or not sediments and fluids are involved in the production of subduction-related magmas. |
| What is found is that for some arcs the concentration of 10Be increases linearly with B/Be ratio. Some arcs like the Central American Arc, the Kurile arc, and the New Britain Arc range to high values of 10Be indicating the incorporation of some component of relatively young oceanic sediment. Other arcs like the Aleutians, Chile, and Kamchatka have low 10Be, but extend to higher values of B/Be. Note that fluids could have rather variable 10Be and B/Be, but the data clearly indicates that some fluid is contributing to the source of subduction-related magmas, consistent with other information we have examined. |  |
Origin Over the last fifteen years or so we have come to a much clearer picture of the origin of the calc-alkaline suite of rocks that is commonly associated with subduction. We here summarize the current theory based on the information discussed above:
Note that despite what your Physical Geology Textbook said about the origin of these rocks, they do not appear to be generated by melting of the subducted oceanic crust, although subducted crust and sediment do appear to contribute to the source. |
Examples of questions on this material that could be asked on an exam
|